 These ions and the atom of argon are known as isoelectronic. Notice that the three ions have electronic configurations identical to that of inert argon. The charges on the chlorine, potassium, and calcium ions result from a strong tendency of valence electrons to adopt the stable configuration of the inert gases, with completely filled electronic shells. Table 2 compares three ions and a neutral atom. Atoms share electrons and form covalent bonds to satisfy the octet rule. 4.2: Covalent Bonds and the Periodic Table is shared under a CC BY-NC-SA 3.0 license and was authored, remixed, and/or curated by LibreTexts. The atoms in group 6A make two covalent bonds. In Table, the common oxidation numbers in the last column are interpreted as the result of either losing the valence electrons (leaving a positive ion) or gaining enough electrons to fill that valence subshell. Covalent bonds are formed by two atoms sharing electrons. For example, in the H 2O molecule, each H has an oxidation number of +1, and the O is –2. One simple piece of evidence for this is the Noble Gases which form the last column on the right of the periodic table. In molecules, the various atoms are assigned chargelike values so the sum of the oxidation numbers equals the charge on the molecule. An interactive Periodic table can be found Periodic Table of the Elements, LibreTexts. The semimetals lie along a diagonal line separating the metals and nonmetals. The metals are on the bottom left in the periodic table, and the nonmetals are at the top right. These are the valence electrons.įor ions, the valence equals the electrical charge. 1: The Periodic Table Showing the Elements in Order of Increasing Z. We saw that Mendeleev arranged elements with similar periodic properties in the same. Do you notice any patterns in the number of valence electrons The number of valence electrons increases going left to right across a period. These electrons are most distant from the positive nucleus and, therefore, are most easily transferred between atoms in chemical reactions. Electron Configurations: Valence Electrons and the Periodic Table. Valence electrons on the periodic table Bohr models for the first three periods of the periodic table are shown below. Valence electrons, which comprise the valence shell of the atom.įor brevity, many chemists record the electron configuration of an atom by giving only its outermost subshell, like 4 s 1 for potassium or 4 s 2 for calcium. 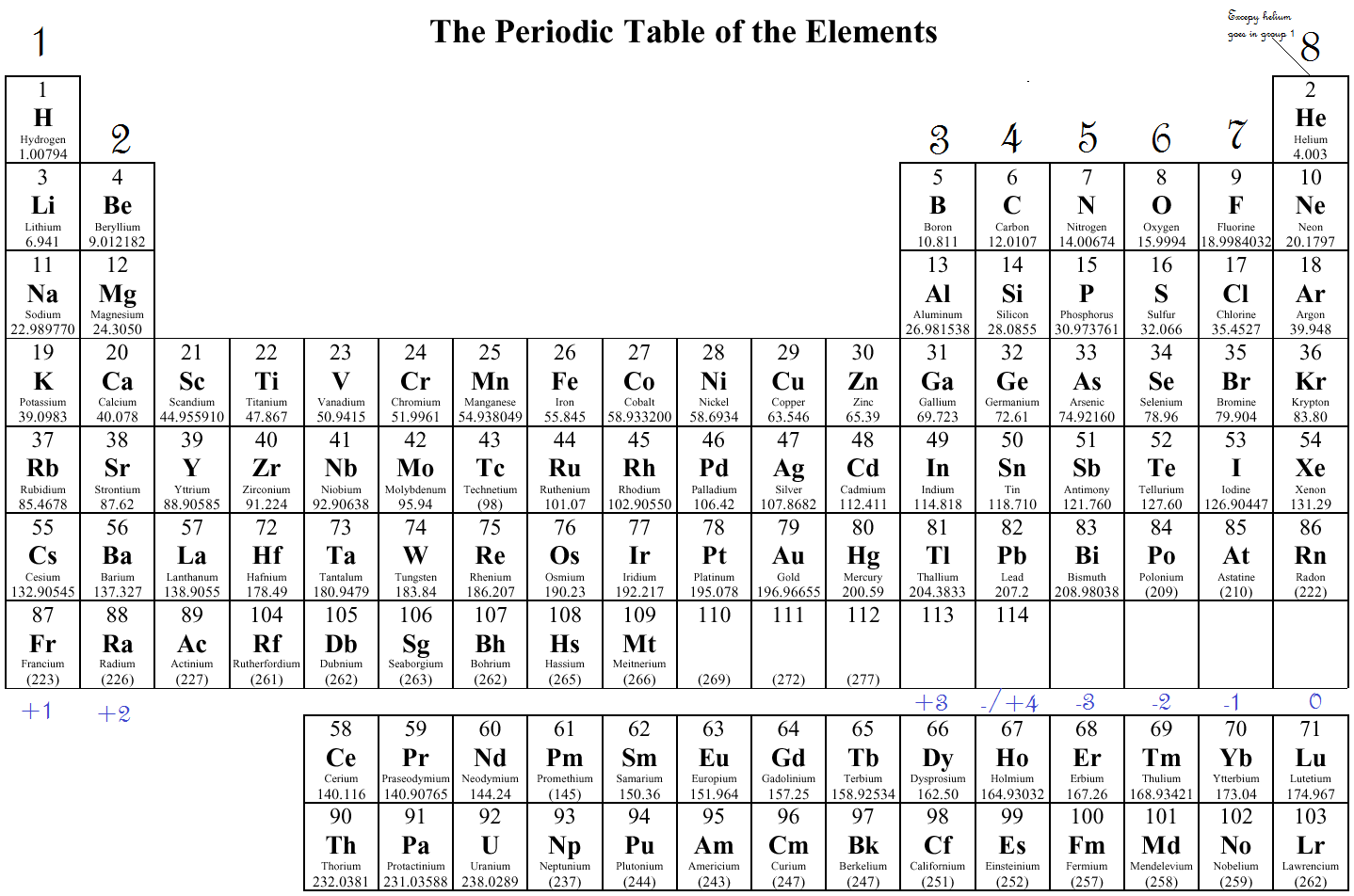
The electrons in the highest numbered subshells are the Study the third column of complete electronic configurations carefully so you understand how electrons are added to the subshell of lowest energy until it reaches its capacity then the subshell of the next energy level begins to be filled. How to determine the number of valence electrons and draw Lewis structures for main group elements starting from the electron configuration. The electronic configuration of an atom is given by listing its subshells with the number of electrons in each subshell, as shown in Tableġ. Groups 312 are transition metals and have 1 or 2 valence electrons. Atoms in a period have the same number of electron shells. An element period is a horizontal row on the periodic table. Atoms in a group share the same number of valence electrons. The electronic formula describes the distribution of electrons over the energy levels that exist in the. An element group is a vertical column on the periodic table. Look at the group that the element is in, as the group number. How many valence electrons does an element have You can use the periodic table to help you determine how many valence electrons an element (specifically, a neutral atom of the element) has. Only the noble gases (the elements on the right-most column of the periodic table) have zero charge with filled valence.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
